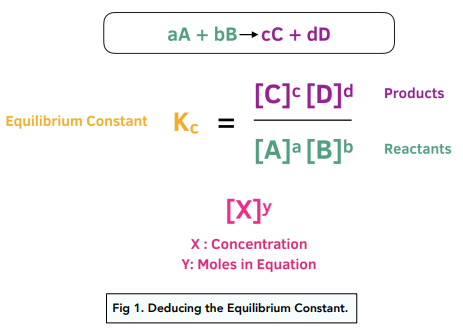
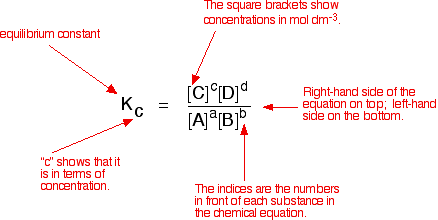
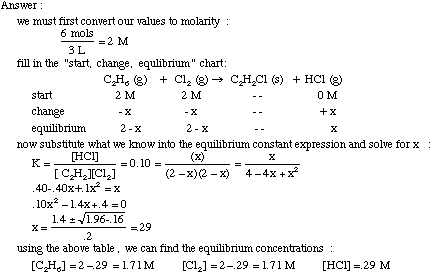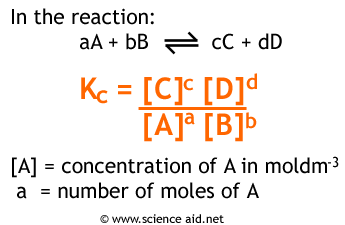Calculate the equilibrium constant for the redox reaction at 25°C. Sr(s) + Mg^(2+) → Sr^(2+)(aq) + Mg(s), - Sarthaks eConnect | Largest Online Education Community

Calculating equilibrium constants from equilibrium concentrations or partial pressures (worked examples) (video) | Khan Academy

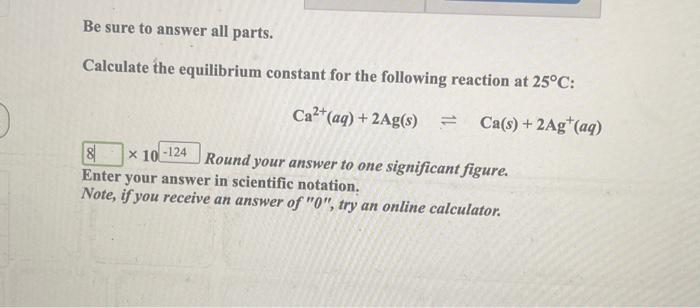
SOLVED: Calculate the equilibrium constant for the following reaction at 25°C: Zn2+(aq) + 2Ag(s) ⇌ Zn(s) + 2Ag+(aq) Round your answer to one significant figure. Enter your answer in scientific notation. Note,