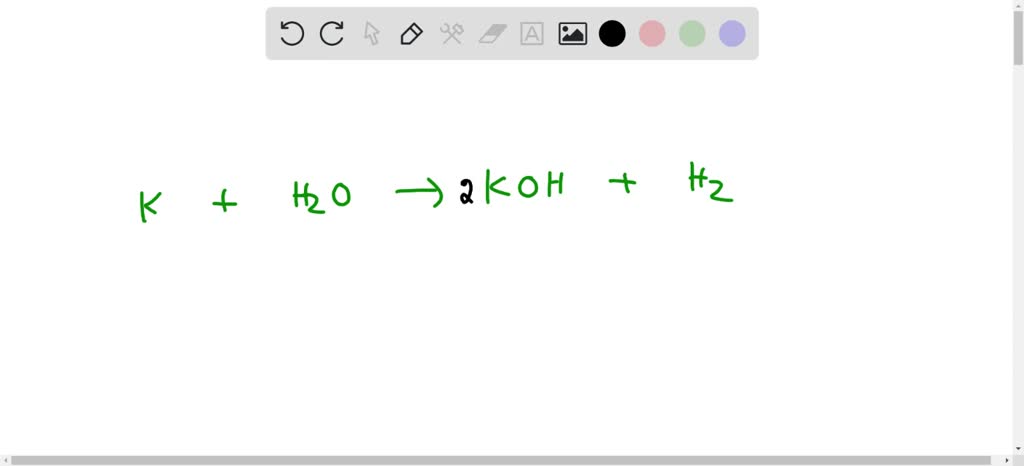
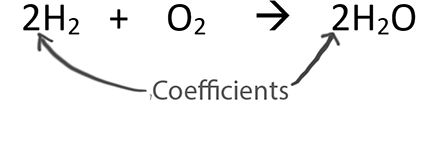Hemp UX ONT DESTIUPS umy Question 2 Complete and balance the following equations: (i) K+H2O (ii) ZnO+C (iii) Mg + O2 → (iv) CaO + H2O → (v) PbO+ H2 -
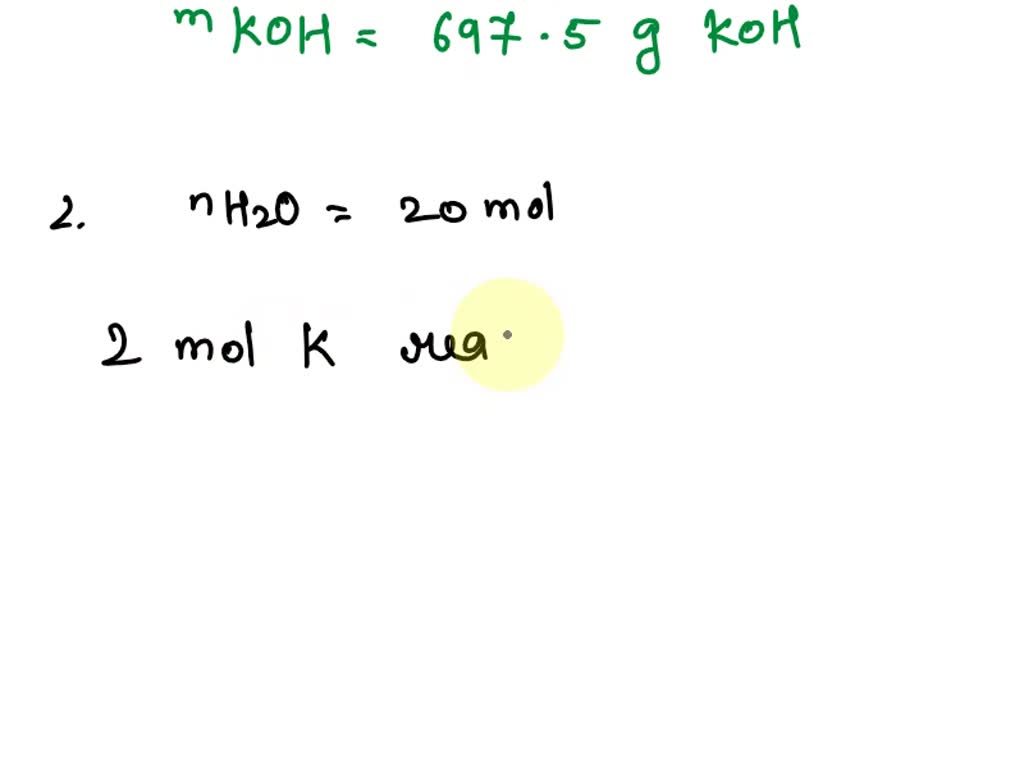
SOLVED: K + H2O –> KOH + H2 1. How many grams of KOH are produced if 224 g of H2O are used? 2. How many moles of K are used if

SOLVED: Balance the following equations. Do not include the states of matter. (a) C + O2 -> CO2 (b) KOH + H3PO4 -> K2HPO4 + H2O (c) N2 + H2 -> NH3 (d) K + H2O -> KOH + H2

SOLVED: Write a balanced net ionic equation for the reaction of potassium with water. a. 2 K(s) + H2O(l) â†' No reaction b. 2 K(s) + 2 H2O(l) â†' 2 K+(aq) +

How to balance K+H2O=KOH+H2|Chemical equation K+H2O=KOH+H2|reaction balance K+H2O=KOH+H2| K+H2O= - YouTube

Oxidation Number method. K+H2O=KOH+H2. Balance the chemical equation by oxidation Number method. - YouTube

Balance KOH + H2SO4 = K2SO4 + H2O (Potassium Hydroxide and Sulfuric Acid) | Balance KOH + H2SO4 = K2SO4 + H2O (Potassium Hydroxide and Sulfuric Acid) Hello Everyone! Welcome back to

SOLVED: Are these equations balanced or unbalanced? H2 + O2 > H2O H2 + O2 > H2O2 K + O2 > K2O Na + Cl > NaCl