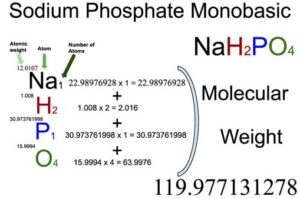
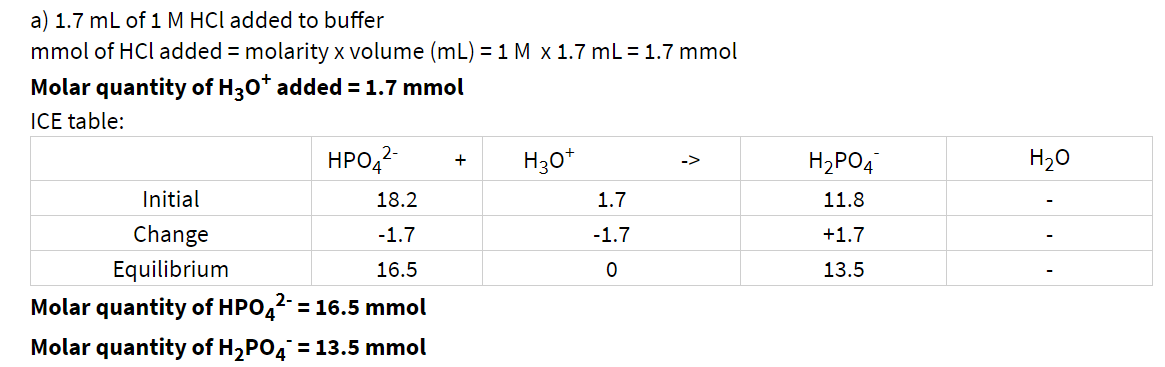SOLVED: And, if necessary, adjust the pH to 7.4 (using either the 0.1 M HCl or 1.0 M NaOH solutions). Adjust the final volume to 100 mL with DDW. Save this solution

Sodium Phosphate Buffer (PH 5.8 To 7.4) Preparation and Recipe - AAT Bioquest | PDF | Buffer Solution | Analysis
How to prepare an 0.1M PBS solution from 1 g of PBS tablet? How much distilled water is required - Quora






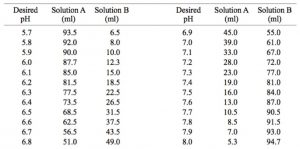


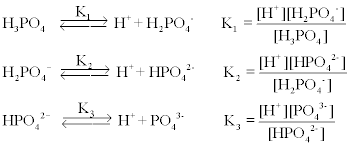


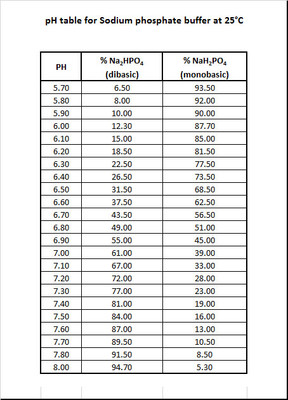
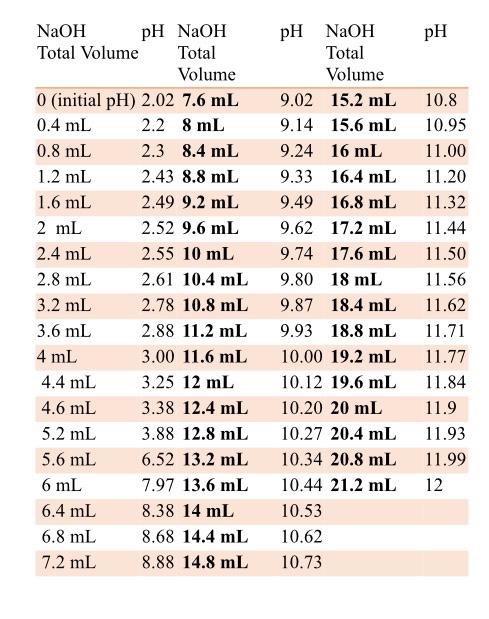
![Sodium Phosphate Dibasic Dihydrate [Na2HPO4.2H2O] Molecular Weight Calculation - Laboratory Notes Sodium Phosphate Dibasic Dihydrate [Na2HPO4.2H2O] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2023/01/sodium-phosphate-dibasic-dihydrate-molecular-weight-calculation-300x186.jpg)